Juha Flinkman put this info together. flinkman@fimr.fi
Hi Jeff & others,
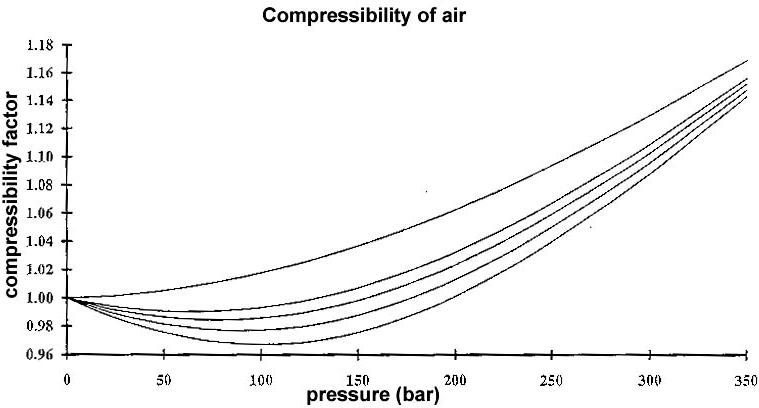
Damn, I forgot to put the legends on that graph: The various curves are AIR at different temperatures: From down
on up they're 270 K, 280 K, 290 K, 300K,and 350 K, or -3deg C, +7deg C, +17 deg C, +27 deg. C, and +77 deg C. I
don't have a Fahrenheit scale handy, but you can do the transforming. Sorry about that. I hope this clarifies things
up a bit
On helium, the compressibility factor at 100 bars/17deg C is ~5%, and at 150 bars/17deg C ~7%, i.e. *more* than
air. This, I think, has to do with ideal gas law basics. With compressed helium, you have lots of hard collisions,
which you're not supposed to have in an ideal gas. In addition to Sychev et al. on air, there's more books for
all gases. I'll be back with those references as soon as I get them. Now, mixing these gases, that's another thing..
I'll put that graph up once more, now that you have the curve legends. Good dives,
Juha
Jeff Bentley
jeffbentley@mindspring.com
My home page
